Non-Small Cell Lung Cancer Market Insights, Epidemiology and Market Forecast-2030
Some of the key facts of the report
1. The total incident cases ofNon-Small Cell Lung Cancer in the 7MM were 484,726 cases in 2017.
2. The total incident cases of NSCLC patients by histology in Germany were observed to be 16,506 cases of squamous cell carcinoma; 26,409 cases of adenocarcinoma and 6,602 cases of large cell carcinoma in 2017.
3. The total diagnosed cases of NSCLC patients by stages in the 7MM were observed to be 86,590 cases of stage I; 31,552 cases of stage II; 56,297 cases of stage IIIA; 65,410 cases of stage IIIB; and 244,876 cases of stage IV in 2017.
Key benefits of the report
1. Non-Small Cell Lung Cancer market report covers a descriptive overview and comprehensive insight of the Non-Small Cell Lung Cancer Epidemiology and Non-Small Cell Lung Cancer market in the 7 MM (the United States, EU5 (Germany, Spain, France, Italy, UK) & Japan.)
2. Non-Small Cell Lung Cancer market report provides insights into the current and emerging therapies.
3. Non-Small Cell Lung Cancer market report provides a global historical and forecasted market covering drug outreach in 7 MM.
4. Non-Small Cell Lung Cancer market report offers an edge that will help in developing business strategies by understanding trends shaping and driving the Non-Small Cell Lung Cancer market.
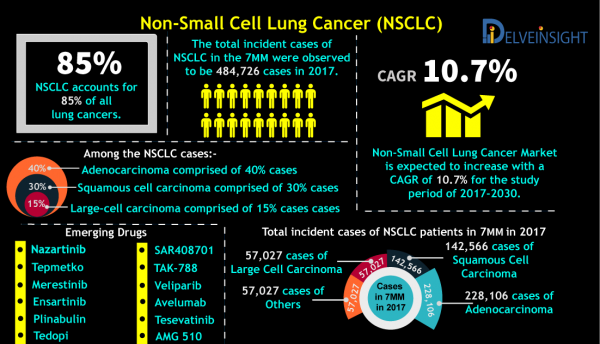
“Non-Small Cell Lung Cancer Market is anticipated to grow at a CAGR of 10.7% for 2017-2030 study period.”
Lung cancer mainly commences in the lungs and may spread to lymph nodes or other organs in the body like the brain. When the cancer cells spread from one organ to another, they are known as metastases. Lung cancer is a significant disease burden on the world and the greatest cause of cancer-related death.
Lung cancer is of two types:- Small Cell Lung Cancer (SCLC) And Non-Small Cell Lung Cancer (NSCLC). NSCLC can be defined as epithelial lung cancer other than SCLC. NSCLC emerges from the epithelial cells of the lung of the central bronchi to terminal alveoli. It is mainly subcategorized into adenocarcinomas, squamous cell carcinomas, large cell carcinomas and several other types that occur less frequently include adenosquamous carcinomas and sarcomatoid carcinomas.
Non-Small Cell Lung Cancer, the most common type of lung cancer that is accounted for approximately 85% of all lung cancers. Among the NSCLC cases, squamous cell carcinoma comprised of 30% cases, adenocarcinoma comprised of 40% cases, and large-cell carcinoma comprised of 15% cases.
There are different types of treatment available for NSCLC; however, mainly ten types of standard treatment are used, which include: Surgery, Radiation therapy, Chemotherapy, Targeted therapy, Immunotherapy, Laser therapy, Photodynamic therapy (PDT), Cryosurgery, Electrocautery, and Watchful waiting.
Chemotherapy is used to stop the growth of cancer cells, either by killing the cells or by stopping their division. Targeted therapy utilizes drugs or other substances to attack specific cancer cells. These therapies usually cause less harm to healthy cells than chemotherapy or radiation therapy. Monoclonal antibodies and tyrosine kinase inhibitors are the two main types of targeted therapy being used to treat advanced, metastatic, or recurrent NSCLC. Immunotherapy uses the patients’ immune system to fight cancer. Radiation therapy uses very high-energy x-rays or other types of radiations to kill cancer cells or keep them from developing.
The launch of the emerging therapies is expected to significantly impact Non-Small Cell Lung Cancer treatment scenario in the upcoming years:-
Drugs covered
– Nazartinib (EGF816)
– Capmatinib (INC280)
– Tepmetko (tepotinib)
– Merestinib
– JNJ-61186372 (JNJ-6372)
– Vemurafenib Plus Cobimetinib
– X-396 (Ensartinib)
– Tedopi (OSE2101)
– Selpercatinib (LY3527723/ LOXO-292)
– SAR408701
– Braftovi (encorafinib) + Mektovi (binimetinib)
– PADCEV (enfortumab vedotin/ASG-22ME)
– Pralsetinib (BLU-667)
– TAK-788: Canakinumab (ACZ885)
– Avelumab (Bavencio)
– Veliparib (ABT-888)
– Sitravatinib (MGCD516)
– Tesevatinib
– Romiplostim
– Cabozantinib
– Sym015
– AMG 510
– INCMGA00012 (MGA012)
– Libtayo (Cemiplimab)
– Bavituximab
– M7824 (Bintrafusp alfa)
And many others
Key Players covered in the report are:-
– Novartis Pharmaceuticals
– Merck KGaA
– Eli Lilly and Company
– Janssen Research & Development
– Hoffmann-La Roche
– Xcovery
– BeyondSpring Pharmaceuticals
– OSE Immunotherapeutics
– Sanofi
– Pfizer
– Astellas Pharma
– Seattle Genetics
– Blueprint Medicines Corporation
– Takeda
– AbbVie
– Kadmon Corporation
– Amgen
– Symphogen
– Regeneron Pharmaceuticals
– Peregrine Pharmaceuticals
– Avid Bioservices
– GlaxoSmithKline
And many others
Table of contents
1. Key Insights
2. Executive Summary of NSCLC
3. SWOT Analysis of NSCLC
4. Non-Small Cell Lung Cancer Market Overview at a Glance
5. NSCLC Disease Background and Overview
6. NSCLC Diagnosis
7. Non-Small Cell Lung Cancer Epidemiology and Patient Population
7.1. The United States Epidemiology
8. EU-5 Epidemiology
8.1. Germany
8.2. France
8.3. Italy
8.4. Spain
8.5. The United Kingdom
9. Japan Epidemiology
10. Non-Small Cell Lung Cancer Current Treatment Practices
11. Guideline of NSCLC
12. Unmet Needs of NSCLC
13. Key Endpoints in NSCLC Clinical Trials
14. Non-Small Cell Lung Cancer Marketed Therapies
14.1. Key Cross
14.2. Rozlytrek (Entrectinib): Hoffmann-La Roche (Genentech)
14.3. Imfinzi (Durvalumab): AstraZeneca
14.4. Opdivo (Nivolumab): Bristol-Myers Squibb
14.5. Tecentriq (Atezolizumab): Genentech/Hoffmann-La Roche
14.6. Keytruda (Pembrolizumab): Merck
14.7. Tafinlar(Dabrafenib)in Combination with Mekinist(Trametinib): Novartis
14.8. Tagrisso(Osimertinib): AstraZeneca
14.9. Lorbrena/Lorviqua (Lorlatinib): Pfizer
14.10. Vizimpro(Dacomitinib): Pfizer
14.11. Alunbrig (Brigatinib): Takeda Pharmaceuticals
14.12. Alecensa (Alectinib): Hoffmann-La Roche
14.13. Vitrakvi (Larotrectinib): Bayer Healthcare
14.14. Portrazza (Necitumumab): Eli Lilly
15. Non-Small Cell Lung Cancer Emerging Therapies
15.1. Nazartinib (EGF816): Novartis Pharmaceuticals
15.2. Capmatinib (INC280): Novartis Pharmaceuticals
15.3. Tepmetko (tepotinib): Merck KGaA
15.4. Merestinib: Eli Lilly and Company
15.5. JNJ-61186372 (JNJ-6372): Janssen Research & Development
15.6. Lazertinib: Yuhan Corporation/ Janssen Research & Development
15.7. Vemurafenib Plus Cobimetinib: Hoffmann-La Roche
15.8. X-396 (Ensartinib): Xcovery
15.9. Plinabulin: BeyondSpring Pharmaceuticals
15.10. Tedopi (OSE2101): OSE Immunotherapeutics
15.11. Selpercatinib (LY3527723/ LOXO-292): Eli Lilly and Company
15.12. SAR408701: Sanofi
15.13. Braftovi (encorafinib) + Mektovi (binimetinib): Pfizer
15.14. PADCEV (enfortumab vedotin/ASG-22ME): Astellas Pharma/Seattle Genetics
15.15. Pralsetinib (BLU-667): Blueprint Medicines Corporation
15.16. TAK-788: Takeda
15.17. Canakinumab (ACZ885): Novartis Pharmaceuticals
15.18. Avelumab (Bavencio): Merck KGaA and Pfizer
15.19. Veliparib (ABT-888): AbbVie
15.20. Sitravatinib (MGCD516): Mirati Therapeutics/Bristol-Myers Squibb/Beigene
15.21. Tesevatinib: Kadmon Corporation
15.22. Romiplostim: Amgen
15.23. Cabozantinib: Exelixis/Ipsen/Takeda
15.24. Sym015: Symphogen
15.25. AMG 510: Amgen
15.26. INCMGA00012 (MGA012): Incyte Corporation/ Zai Lab Limited
15.27. Libtayo (Cemiplimab): Regeneron Pharmaceuticals
15.28. Bavituximab: Peregrine Pharmaceuticals/ Avid Bioservices, Inc
15.29. M7824 (Bintrafusp alfa): GlaxoSmithKline/Merck KGaA
16. NSCLC Seven Major Market Analysis
17. PD-L1—Market Size
18. BRAF Mutation—Market Size
19. c-MET Mutation—Market Size
20. EGFR Mutation—Market Size
21. ALK-Mutation—Market Size
22. Market Access and Reimbursement of NSCLC Therapies
23. Market Drivers of NSCLC
24. Market Barriers of NSCLC
25. Appendix
26. DelveInsight Capabilities
27. Disclaimer
28. About DelveInsight
About DelveInsight
DelveInsight is a leading Business Consultant, and Market Research Firm focused exclusively on life sciences. It supports pharma companies by providing end to end comprehensive solutions to improve their performance.
Contact us:
[email protected]
+919650213330
SOURCE DelveInsight
Media Contact
Company Name: DelveInsight
Contact Person: Priya Maurya
Email: Send Email
Phone: +919650213330
City: Albany
State: New York
Country: United States
Website: https://www.delveinsight.com/